Diamond as a Plasma-Facing Protective Coating in Fusion Reactors
Diamond as a Plasma-Facing Protective Coating in Fusion Reactors
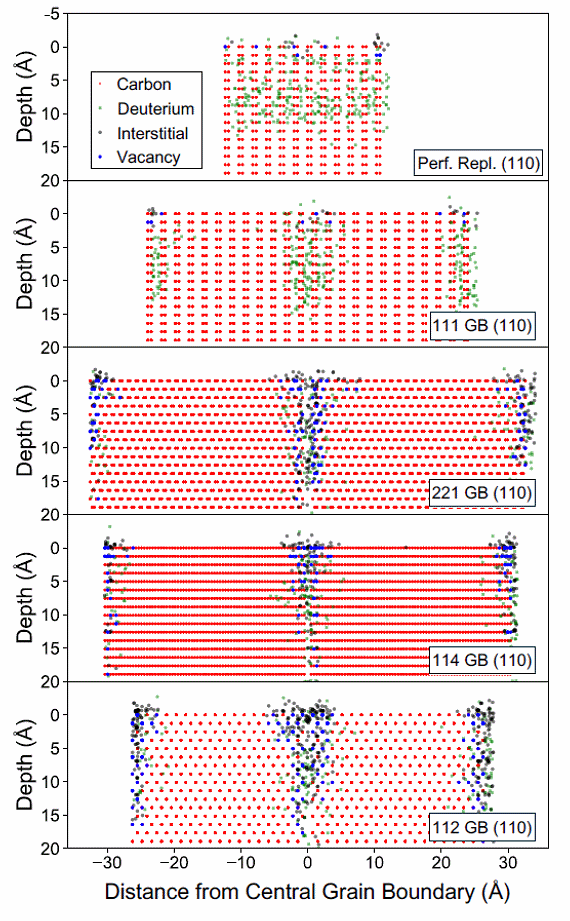
The plasma-facing material (PFM) is the material that lines the inside of the first wall and divertor of a nuclear fusion reactor. The primary role of a PFM is to protect more delicate components of the reactor from high thermal loads and low-energy ion irradiation. Such materials will also need to endure high fluences of fast neutrons, but typically will do little to protect the other components of a wall. Diamond�s intrinsic hardness, excellent thermal properties, resistance to radiation damage, semiconducting properties and transparency, make it a material of interest for numerous applications within fusion energy. These applications include PFMs, windows, hydrogen permeation barriers, as well as neutron and tritium detectors. All of these potential applications involve the interaction of diamond with hydrogen isotopes on some level. The two most crucial things to understand are whether the diamond will etch away under the flux of high energy H ions, i.e. what will be its lifetime, and will it absorb tritium (the main fuel source for fusion) and retain it within the bulk diamond or in grain boundaries? Such captured tritium will not be available for gas-phase fusion, and so reduce the efficiency of the process. Knowing how much tritium is absorbed and retained by CVD diamond is critical to understanding whether diamond can be used as a PFM. |
 Plasma-facing wall in JET. [Image: UKAEA] |
For the TDS experiments, both undoped and boron-doped diamond samples were exposed to deuterium ions in DELPHI at UKAEA Culham Science Campus. Deuterium is easier to handle than tritium and can be distinguished from normal H easily, while still remaining a good model for tritium. D2 retention in the diamond was determined with either TDS or Nuclear Reaction Analysis (NRA). Undoped-diamond results suggested a similar number of available hydrogen/deuterium binding sites in all samples, and the amount of D2 desorbed is linked to the initial hydrogen content. The inclusion of boron dopants increased retained deuterium. The most significant increases seen in this work are thought to be a result of an increase in surface roughness of the unpolished films. Another peak that is not present for undoped samples was observed at 700 K. This is thought to be a direct result of the inclusion of B through either the formation of boron-hydrogen complexes or the inclusion of boron adding disorder to the atomic structure. |
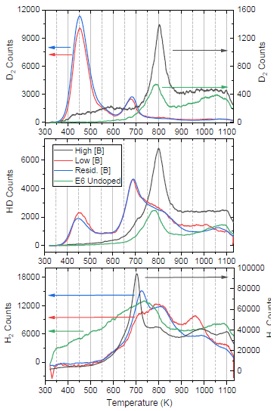 TDS spectra for masses 2, 3 and 4 attributed to H2, HD and D2, respectively, for three diamond samples grown with different levels of gas-phase B concentrations and a undoped reference sample. A ramp rate of 0.167 K/s was used. Where there are large discrepancies in counts, two axes have been used, coloured arrows indicate the axes corresponding to each spectrum. |
Related Papers
- J.A. Pittard, N.A. Fox, A. Hollingsworth, M.Y. Lavrentiev, A. Wohlers and Y. Zayachuk, "Deuterium retention in CVD diamond: Combined experimental and computational study", Fus. Engineer. and Des., 188 (2023) 13403. [doi: 10.1016/j.fusengdes.2022.113403]
- J.A. Pittard, M.Y. Lavrentiev, N.A. Fox, "Simulated bombardment of diamond with hydrogen isotopes", IEEE Trans. Plasma Sci., 52 (2024) 3674-3679. [doi: 10.1109/TPS.2024.3435521]
- J.A. Pittard, M.Y. Lavrentiev and N.A. Fox, "Simulated hydrogen diffusion in diamond grain boundaries", Diamond Relat. Mater. 149 (2024) 111665. [doi: 10.1016/j.diamond.2024.111665]